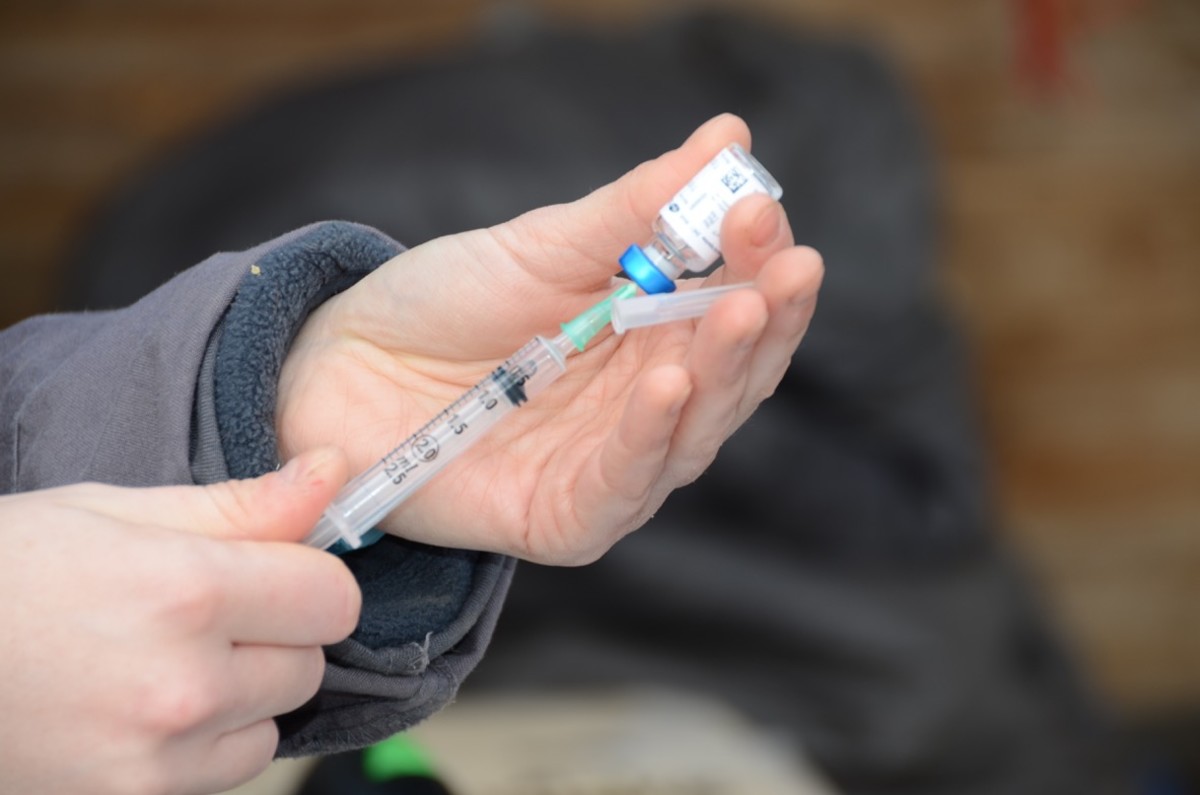
A research paper was published titled, “Equine influenza vaccination in the UK—current practices may leave horses with suboptimal immunity.” The paper was authored by Wilson, A.; Pinchbeck, G.; Dean, R.; and McGowan, C.
This research survey of equine veterinarians aimed to describe current vaccination practices advised by veterinarians treating horses in the United Kingdom (UK) and compare those practices with manufacturer datasheets and current guidelines.
A survey completed by 304 UK equine vets working with a variety of horse populations identified that the recommendations given to owners by the majority of respondents (92%) were not consistent with manufacturer datasheet guidelines.
Vaccination guidelines from the British Horseracing Authority and the Fédération Equestre Internationale were most commonly used.
Variation was also identified in vaccine protocols for competition and non-competition horses, with 57% of respondents reporting variation in advised “booster” frequency. This most commonly consisted of advising a 6-month vaccination schedule in competition horses and annual vaccination in non-competition horses.
Adverse events following vaccination in the previous year to the survey were encountered by 66% of respondents, representing 2,760 adverse events in total. Only 526 (19%) of reactions were reported to the Veterinary Medicines Directorate. The most common reactions were transient, including stiffness (n = 931), localized swelling (n = 835), lethargy (n = 559) and pyrexia (n = 355). A large proportion (86%) of respondents reported vaccine hesitancy from horse owners. This was most commonly due to perception of over-vaccination, cost and concerns regarding adverse events.
Bottom line: A discrepancy exists between datasheet guidelines and those issued by industry governing bodies. Non-compliance with the datasheet guidelines is common, risking suboptimal immunity to equine influenza.
You can purchase access or long in to access the full article from the Wiley online library.


![[Aggregator] Downloaded image for imported item #19723](https://s3.amazonaws.com/wp-s3-equimanagement.com/wp-content/uploads/2026/04/31141904/EDCC-Unbranded-28-scaled-1-768x512.jpeg)
![[Aggregator] Downloaded image for imported item #19767](https://s3.amazonaws.com/wp-s3-equimanagement.com/wp-content/uploads/2026/05/31141830/EDCC-Unbranded-26-scaled-1-768x512.jpeg)
![[Aggregator] Downloaded image for imported item #20008](https://s3.amazonaws.com/wp-s3-equimanagement.com/wp-content/uploads/2026/05/29132757/EDCC-Unbranded-15-scaled-1-768x511.jpeg)