 Credit: Arnd Bronkhorst Photography
Credit: Arnd Bronkhorst PhotographyThe American Association of Equine Practitioners (AAEP) defines “core vaccinations” as those that provide protection against diseases that are endemic to a region; those with potential public health significance; those required by law; those that protect against diseases that are virulent/highly infectious; and/or those that protect against the risk of severe disease.
Core vaccines have clearly demonstrated efficacy and safety, exhibiting a high enough level of patient benefit coupled with a low enough level of risk to justify their use in the majority of patients. The following equine vaccines are identified as “core” by the AAEP: Eastern and Western equine encephalitis (EEE and WEE), tetanus, rabies and West Nile virus (WNV).1
Every vet’s goal regarding these core vaccines should be to educate every client in order to reach every horse and have it vaccinated properly. Articulating the dangers of these diseases to clients can be productive in encouraging compliance with vaccination recommendations. Being aware of current research and regional cases can strengthen your case. Here, we will help update you on the causative organisms and provide you with details that you can relate to your clients.
The Pathogens
Eastern and Western equine encephalitis and West Nile virus are all arboviruses that are transmitted by blood-feeding insects or ticks and can cause fatal neurologic disease in horses. All of these viruses are zoonotic, and thus have public health implications that should be communicated to your clients.
EEE, WEE and WNV can each be spread from birds to humans via mosquitoes. However, horse-to-horse or horse-to-human transmission by mosquitoes does not occur because the level and duration of viremia in a horse ill with EEE, WEE or WNV is insufficient to infect mosquitoes that might feed on the infected equine.
Horses infected with EEE, WEE or WNV are indicators that the virus is active in a geographic area and that vigilance in reducing mosquito habitat is necessary to minimize risk to humans. Viral encephalitis in an animal is a reportable disease in most states for this reason.
Although there have been no cases in the U.S. for 40 years, Venezuelan equine encephalomyelitis (VEE) has the potential to be a much more serious disease than the aforementioned viruses, as it causes a high level of viremia and thus can be transmitted from horses to mosquitoes to humans. VEE is a reportable foreign animal disease.
Tetanus generally is a fatal disease caused by a potent neurotoxin produced by the anaerobic, spore-forming bacterium, Clostridium tetani. Cl. Tetani organisms are present in the intestinal tract and feces of animals (including horses) and humans; they are abundant as well as ubiquitous in soil. Spores of Cl. tetani survive in the environment for many years, resulting in a continuing risk of exposure for both horses and people. Tetanus is not a contagious disease; it is the result of Cl. tetani infection of puncture wounds, open lacerations, surgical incisions and exposed tissues such as the umbilicus of foals and the reproductive tract of the postpartum mare (especially in the event of dystocia or retained placenta).
Rabies virus causes an infrequently encountered, but invariably fatal, neurologic disease. Although all species of mammals are susceptible to rabies, only a few species are important as reservoirs. In the United States, several distinct rabies virus variants have been identified in terrestrial mammals, including raccoons, skunks, foxes and coyotes. In addition to these terrestrial reservoirs, several species of bats are also reservoirs for rabies.2
While the incidence of rabies in horses is low, the disease has considerable public health significance. Exposure occurs through the bite of an infected (rabid) animal. Bites to horses occur most often on the muzzle, face and lower limbs. The virus migrates via nerves to the brain, where it initiates rapidly progressive, invariably fatal encephalitis.
Epidemiology of the Diseases
Eastern/Western Encephalitis (EEE/WEE)
In the United States, the distribution of EEE has historically been primarily in the Eastern, Southeastern and some Southern states, but disease incidence has also been reported in the upper Midwestern states of Ohio, Michigan and Wisconsin. In recent years, multiple cases of EEE have been reported in humans as well as in unvaccinated horses, offering clear evidence that this virus is still circulating.
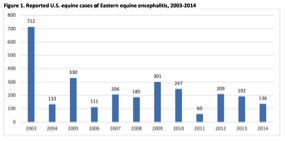
Reported US equine cases of EEE, 2003-20143
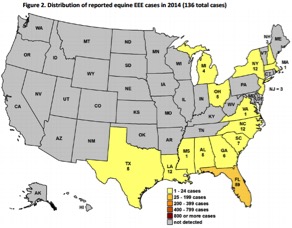
Distribution of reported equine EEE cases in 2014
(136 total cases)4
WEE is spread primarily by mosquitoes and occasionally by small, wild mammals. In the warm, moist environments of the West Coast, cycles of wild bird/mosquito interactions allow the virus to remain endemic. No cases of bird transmission of the disease have been reported, making mosquitoes the primary vector and birds simply reservoirs.
Epidemic outbreaks in the equine population generally precede human epidemics of WEE.5 Outbreaks have been recorded in the Western and Midwestern states in the past, most notably in 1987, when equine cases were identified in 11 Southwestern, Plains and Rocky Mountain states as well as in Canada.6 Variants of WEE have also caused sporadic cases in the northeast and southeast United States.
Transmission of EEE and WEE from wild birds or rodents, which serve as natural reservoirs, through mosquitoes to horses varies from year to year, with changes in distribution of insect vectors and reservoirs. EEE activity in mosquitoes and birds, and resultant disease in humans and equines, continues to cause concern along the Gulf, Southern and East Coasts.
EEE and WEE are very similar clinically in horses, although the course of EEE might be shorter. EEE is the most virulent for horses, with mortality approaching 95%. WEE has a lower mortality rate of 25-50%. Epidemiological evidence indicates that young horses are particularly susceptible to disease caused by EEE virus. Acute clinical signs of EEE and WEE are nonspecific, and include mild to severe fever (103-106° F) that lasts 24 to 28 hours, poor appetite and stiffness. Many cases of WEE do not progress beyond this point.
With EEE, progression is nearly certain. As the disease progresses, clinical signs include propulsive walking, depression, and somnolence to aggression and excitability. Some horses can become frenzied after any stimulation. Later signs indicating increased severity of brain dysfunction include head pressing, propulsive walking, blindness, circling, head tilt and facial and appendicular (limb) muscle fasiculations.
Paralysis of the pharynx, larynx and tongue are common. Defecation and urination become difficult; some animals develop diarrhea or constipation or have significant weight loss. Complete paralysis and death is often preceded by recumbency for one to seven days. Periods of excitement or intense pruritus have been reported, and laterally recumbent animals sometimes have a characteristic paddling motion. This final stage might appear two to four days after the first signs appear. In some cases, horses with EEE might show no clinical signs before dying. Animals that are comatose rarely survive. If animals survive, they may show gradual improvement of function over weeks to months.
Treatment for EEE or WEE is limited to supportive care. Horses will have immunity for up to two years after infection, but might have permanent neurologic damage.
In equines, EEE and WEE can be diagnosed by serology, particularly by the presence of antibodies in an IgM antibody-capture ELISA, or a four-fold rise in titer in the plaque reduction neutralization (PRN) test. In horses with a history of vaccination, a four-fold rise in neutralizing antibody titer from acute and convalescent blood samples (taken two to four weeks apart) is diagnostic.
PCR (polymerase chain reaction test, which detects virus DNA) tests for EEE are available. Unlike in humans, cerebrospinal fluid (CSF) is not considered more reliable for detecting EEE-specific IgM than serum. A presumptive diagnosis can be obtained with a high titer in a single sample from an unvaccinated horse, particularly when a combination of serological tests is used. Because viremia usually occurs early in the infection (before the onset of neurological signs), blood is unlikely to contain EEE in affected horses. This virus might be isolated from the brain after death, as the amount of virus in this tissue is often high, but it can disappear if the illness is prolonged. It can also sometimes be found in extra-cranial tissues such as the liver or spleen. Virus isolation is rarely successful in WEE-infected horses.7
Annual vaccination with the available formalin inactivated adjuvanted whole virus products must be completed prior to vector season in the spring. In locations of high risk or for horses with limited immunity, more frequent vaccination or appropriately timed vaccination is recommended. In areas where mosquitoes are active year-’round, many vets vaccinate horses at four- to six-month intervals.
West Nile Virus

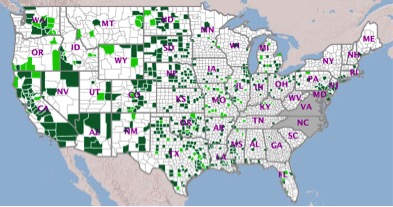
Cumulative West Nile Virus Disease Cases as of October 27,
2015 – Human and Non-Human8
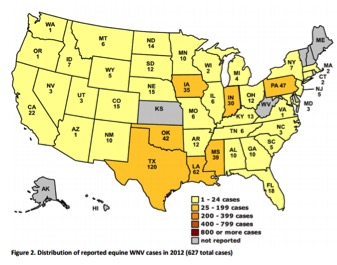
Distribution of WNV cases in 20129
West Nile virus has a much lower mortality rate (33%) than EE, but it typically causes more cases of arbovirus encephalitis each year in U.S. horses. Since 1999, more than 25,000 U.S. horses have developed WNV encephalitis, with a high of 15,257 horses affected in 2002. Horses represent 96.9% of all reported non-human cases of WNV disease in mammals.
The virus is present in the continental U.S., most of Canada and Mexico. The virus is transmitted by mosquitoes from birds, which serve as reservoir hosts, to mammals. Variability in WNV transmission is affected by geography, as bird and mosquito populations result in regional differences in virulence of the virus; therefore, no prediction can be made regarding future trends in local activity of the viruses. Horses and humans are considered to be dead-end hosts for WNV; the virus is not directly contagious from horse to horse or horse to human because infected horses do not circulate a significant amount of virus in their blood.
The clinical signs and course of the disease are highly variable. Not all horses infected with the virus develop the disease, and a few horses die without showing signs of illness. Initially, the horse with WNV might show discomfort, anxiety, lameness, low-grade fever and/or malaise. Neurologic signs can include muzzle twitching, impaired vision, aimless wandering, head pressing, circling, inability to swallow, irregular gait, trembling, lack of coordination, weakness, muscle contractions, paralysis, convulsions and death. Horses can become abnormally sensitive to sights, sounds or touch.
Treatment can include serum or plasma products containing antibodies to the virus, but supportive care is important. Anti-inflammatory drugs, including non-steroidal drugs and/or corticosteroids, are often used. Most recovered horses return to normal function within one to six months. As many as 40% of horses that survive acute illness still exhibit gait and behavioral abnormalities six months later.
The most reliable test for WNV diagnosis in clinically ill horses is the IgM capture ELISA. Although other serological tests might be used to diagnose WNV, the IgM antibody rises quickly after exposure, but is relatively short-lived, so IgM capture ELISA can confirm recent exposure to the virus. A positive result indicates infection likely occurred within the previous three months. Data indicates that little IgM is detected as a result of recent vaccination. There is an inactivated whole WNV vaccine, non-replicating live canary pox recombinant vector vaccine and inactivated flavivirus chimera vaccine. Annual vaccination in the spring, prior to the onset of the insect vector season, is recommended. In areas with year-’round mosquito populations or for horses with limited immunity, more frequent vaccination is indicated.
Tetanus
Tetanus toxemia is caused by a specific neurotoxin produced by Clostridium tetani in necrotic tissue. Almost all mammals are susceptible, although dogs and cats are more resistant than other domestic or lab mammals. Birds are resistant. Horses and lambs seem to be the most sensitive of all species, with the possible exception of people. Although tetanus is worldwide in distribution, there are some areas, such as the northern Rocky Mountain section of the U.S., where the organism is rarely found in the soil.
The spores of Cl. tetani are unable to grow in normal tissue or in wounds if the tissue remains well oxygenated. Bacterial multiplication occurs when a small amount of soil or a foreign object is introduced and tissue necrosis occurs under an anaerobic condition. The bacteria remain localized in the necrotic tissue at the original site of infection and multiply. As bacterial cells undergo autolysis, the potent neurotoxin is released. Toxin is absorbed by the motor nerves in the area and travels retrograde up the nerve tract to the spinal cord.
The toxin causes spasmodic, tonic contractions of the voluntary muscles by interfering with the release of inhibitory neurotransmitters from presynaptic nerve endings. If more toxin is released at the site of the infection than the surrounding nerves can take up, the excess is carried off by the lymph to the bloodstream and to the CNS, where it causes descending tetanus.
Even minor stimulation of the affected animal can trigger characteristic tetanic muscular spasms. The spasms might be severe enough to cause bone fractures. Spasms affecting the larynx, diaphragm and intercostal muscles lead to respiratory failure. Involvement of the autonomic nervous system results in cardiac arrhythmias, tachycardia and hypertension.10
The incubation period varies from one to several weeks, but it usually averages 10–14 days. Localized stiffness, often involving the masseter muscles and muscles of the neck, the hind limbs, and the region of the infected wound, is seen first; general stiffness becomes pronounced about a day later, and tonic spasms and hyperesthesia become evident.
Spasms of head muscles cause difficulty in prehension and mastication. The horse’s ears are held erect, the tail stiff and extended, the anterior nares dilated, and the third eyelid prolapsed. Walking, turning and backing are difficult. Spasms of the neck and back muscles cause extension of the head and neck, while stiffness of the leg muscles causes the animal to assume a “sawhorse” stance. Sweating is common. General spasms disturb circulation and respiration, which results in increased heart rate, rapid breathing and congestion of mucous membranes.
When administered in the early stages of the disease, tranquilizers or barbiturate sedatives in conjunction with 300,000 IU of tetanus antitoxin IV bid have been effective in the treatment of horses. Nursing care in a quiet, darkened box stall is essential. Slings might be useful for horses having difficulty standing or rising.
Vaccines currently available for tetanus are formalin-inactivated, adjuvanted toxoids. Tetanus toxoid is a potent antigen that rapidly induces strong serological responses, and reliably prevents the development of tetanus.
Rabies
After being bitten by an infected animal, the victim’s peripheral nerves transmit the virus to the brain, where it initiates rapidly progressive, invariably fatal encephalitis. The incubation period averages two to nine weeks, but might be as long as 15 months. In the U.S., cats have remained the most frequently reported rabid domestic animal at 53%, followed by dogs (19%), cattle (18%), and horses and mules (7%). In 2013 there were 31 U.S. cases of rabid horses and mules.11
The most commonly reported clinical signs in horses include colic, lameness, ataxia, paralysis, urinary incontinence, muscle tremors, fever, depression, aggressiveness, hyperesthesia and seizures. Most rabid horses exhibit some degree of hyperesthesia, fever and neurological signs at some point during the course of the disease.
While some horses exhibit intermittent or continuous signs of aggression, most are depressed or stuporous. Some can become anorectic and refuse to drink, while others will continue to eat and drink until shortly before death. Obscure lameness and posterior ataxia are common early signs. Progression of the disease is rapid, with death occurring three to five days following the onset of clinical signs. Prior to death, most horses will become recumbent with convulsions and/or a comatose state and violent thrashing. Rabies is always fatal.
Diagnosis of rabies requires tissue from two sites, preferably the brain stem and/or cerebellum. Detection of rabies virus from any part of the affected brain is possible, but in order to rule out rabies, the test should include two tissues. In the U.S., the results of a rabies test are typically available within 24 to 72 hours.
Rabies vaccines are inactivated tissue culture derived products and induce a strong serologic response after a single dose.
The Business of Vaccinations
The AAEP Vaccination Guidelines state that the availability of licensed vaccine products combined with an inability to eliminate risk of exposure justifies immunization against EEE, WEE, tetanus, rabies and West Nile virus for all horses residing in or traveling to North America.
Equine veterinarians have a strong opportunity to provide guidance and education to their clients about these serious diseases. You can help your clients understand why vaccination is important through many channels: newsletters, brochures, farm visits, seminars, “on-hold” messages and social media. Use the information in this article to share the risks of disease and the benefits of vaccination.
The 2012 AAEP Horse Owner-Trainer Veterinary Services Survey revealed that vets are highly valued and trusted, with a strong majority of the respondents saying that they value their vets’ opinions more than anything they might read on the Internet. These owners have such a level of trust that 89% report that they “always” or “almost always” take their primary vets’ advice when it comes to vaccinations.
Despite the drumbeat of doom that whispers that vaccines are a lost cause in practice, do not believe it!
The owners surveyed by AAEP repeatedly revealed that they want vets who exhibit compassion; explain medical and husbandry issues in an understandable way; seek and respect the opinion of the owner; and take time with them.
Providing annual health exams when administering core vaccinations is an excellent way to form these bonds; this personal approach is a clear contrast to the quick vaccine visits that so many clients have experienced in the past.
Vaccines can be considered commodities because they are widely available from multiple sources, the provider of the service doesn’t frequently affect the outcome, and it can sometimes be hard to differentiate value between providers. But vaccines don’t have to be a loss leader.
Make personalized health recommendations for each of your patients a front-and- center part of the services you offer. Clients value personal, relationship-based service from their vets, tailored individually for their horses. You are the person best positioned to give them the high-quality medical services and advice that they want. An annual or biannual farm visit for each horse in your practice provides an occasion for a physical exam, body condition scoring, dietary review, dental exam and an airing of owner concerns. Careful attention to each owner’s animal is highly valued.
Whether you wrap core vaccinations into an annual health exam or simply make the process of annual vaccination convenient, economical and personalized for your clients, make vaccines the foundation of your patients’ health maintenance.
References
1. Core Vaccination Guidelines. http://www.aaep.org/info/core-vaccination-guidelines. Accessed 10/30/15.
2. Wildlife Reservoirs for Rabies. Accessed 10/31/15. http://www.cdc.gov/rabies/exposure/animals/wildlife_reservoirs.html
3. USDA 2014 Summary of Eastern Equine Encephalitis Cases in the United States https://www.aphis.usda.gov/animal_health/downloads/animal_diseases/2014_eee_annual_final.pdf
4. Ibid
5. Western Equine Encephalitis. Accessed 10/31/15. http://emedicine.medscape.com/article/233568-overview
6. Epidemiologic Notes and Reports Western Equine Encephalitis — United States and Canada, 1987. Accessed 10/30/15. http://www.cdc.gov/mmwr/preview/mmwrhtml/00000983.htm
7. Eastern, Western and Venezuelan Equine Encephalomyelitis. Accessed 10/30/15. http://www.cfsph.iastate.edu/Factsheets/pdfs/easter_wester_venezuelan_equine_encephalomyelitis.pdf
8. http://diseasemaps.usgs.gov/mapviewer/
9. Equine West Nile Virus. Accessed 11/2/15. https://www.aphis.usda.gov/vs/nahss/equine/wnv/2012_equine_west_nile_virus_final.pdf
10. Tetanus. Accessed 10/31/15. http://www.merckvetmanual.com/mvm/generalized_conditions/clostridial_diseases/tetanus.html
11. Rabies in the US. Accessed 10/31/15. http://www.cdc.gov/rabies/location/usa/surveillance/domestic_animals.html








