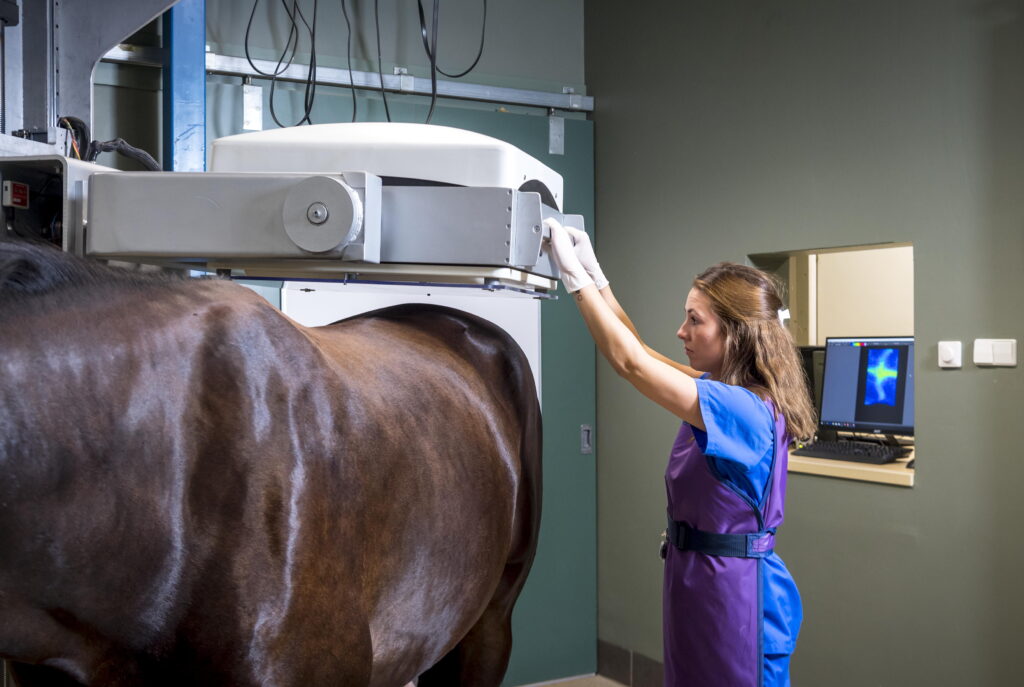
Radiographs were introduced to veterinary medicine about 100 years ago, and they remain the cornerstone of diagnostic imaging in equine patients. It wasn’t until the 1970s that scintigraphy was introduced, followed a decade or so later by ultrasonography. Computed tomography (CT) and magnetic resonance imaging (MRI) were introduced in the 1990s and have since exploded in terms of clinical availability and utility. Lastly, positron emission tomography (PET) was first performed on a horse in 2015 and has recently become available in several clinical practices in the United States. Even though traditional radiographs are still the primary go-to diagnostic technique for many conditions, particularly lameness, our diagnostic capabilities have benefitted immensely from the advent of advanced imaging. MRI, CT, nuclear scintigraphy and PET are now available to many practitioners and their clients.
As pointed out by Kathryn Wulster Bills, VMD, DACVR, DACVR-EDI, from the University of Pennsylvania’s New Bolton Center, “Having more diagnostics means being able to make more specific diagnosis, not only identifying the injured structure but also the degree of current ‘activity’ of injury. These tools ultimately allow us to perform more specific treatments and determine which treatments work best because we decrease the amount of confounding information as to the cause of lameness.”
This article reviews how each of the advanced imaging modalities (CT, MRI, nuclear scintigraphy and PET) are performed. We will also outline the advantages and limitations of their use. Some suggestions on how and when to fit these imaging modalities into your patients’ diagnostic workups are also provided.
CT for Equine Patients
CT is a computerized X-ray imaging procedure that involves aiming a narrow beam of X-rays at the patient and quickly rotating that beam around the body, generating tissue density data for a specific body region. The computer reconstructs this information to create cross-sectional images or “slices.” The volume data can also be displayed as a 2D rendering of a 3D image. After collecting a number of successive slices, the computer digitally stacks them together to form a three-dimensional image of the patient. These images—either the slices called tomographic images or the 3D image—give the clinician more detailed information than conventional X-rays.
“Radiologists and vets that look at a lot of imaging prefer to look at the transverse slices,” said Wulster Bills. “The 3D reconstruction can be very helpful to demonstrate pathology to owners or for surgical planning in cases of complex fracture configurations.”
Advantages
Horses can be imaged using standing sedation, and acquisition times are short, less than one minute per scan once the patient is sedated and positioned appropriately.
“If performed under general anesthesia, limbs can be imaged up to the shoulder or hip depending on the machine,” said Sue Dyson, MA, Vet MB, PhD, DEO, DCVSMR, FRCVS.
Disadvantages
This modality often is limited to the distal limbs, head and neck depending on the CT machine’s aperture.
Some CTs provide helpful information regarding the soft tissues (specifically fan beam CT coupled with intra-arterial contrast administration), but the contrast resolution is still less than with MRI. Thus, the evaluator might not be able to discern individual soft tissue structures, and they will not be able to differentiate cartilage from joint fluid without injection of contrast material within the joint.
Wulster Bills noted, “Radiation exposure of personnel needs to be considered with standing CT systems.”
MRI for Equine Patients

This is a non-invasive imaging technology that acquires images in three planes to produce two-dimensional images. The anatomic region of interest is placed inside a strong magnetic field that forces the protons in those tissues to align. A radiofrequency current is then pulsed through the patient, stimulating the protons and forcing them to spin off their previous alignment. When the radiofrequency is turned off, the protons realign within the magnetic field, releasing energy while doing so. The amount of energy released differs depending on the chemical composition of the tissue. Thus, different tissue types (bone, soft tissue, blood) will produce different signals appreciable on the images. Inflamed tissue, hemorrhage, fibrosis and necrosis of soft tissues can all be identified on MRI.
Two different types of MRI are available: low-field and high-field. This refers to how strong the magnet is. In equine practice for standing patients, only low-field magnets can be used because of their size and not being able to make the unit small enough to fit a standing patient’s limb inside.
“The Hallmarq low-field magnet can also be used with the horse under general anesthesia,” said Dyson. “It was designed to permit horses to be examined standing to avoid the risks of anesthesia and potentially to make the procedure quicker and cheaper. In reality, because of movement problems, image acquisition may take considerably longer and therefore if charged appropriately may not be cheaper.”
She added, “Image resolution is infinitely superior with high-field MRI, slice thickness is reduced, a larger area can be imaged more quickly, and the problems of movement are reduced but not eliminated because limbs can move in synchrony with respiration.”
A variety of examinations can be performed via MRI.
“T1 weighted and T2 weighted images vary in their echo time and various other image acquisition parameters,” said Dyson. “In essence, T2 weighted and STIR images detect signs of more active inflammation/necrosis in soft tissues than T1 weighted.”
For example, high signal intensity in an unhealed, deep digital flexor tendon injury seen in STIR and T2 weighted images might resolve over time, but residual high signal intensity might persist in a T1 weighted image reflecting fibrosis or other degenerative type change (e.g., chondroid metaplasia).
“Normal tendon healing also remains visible on T1w images because tendons and ligaments mostly heal in a short random fiber pattern which, due to an MRI artifact called magic angle, leads to persistent T1 weighted increased signal,” noted Wulster Bills.
“We also have gradient echo sequences and fast spin echo sequences with the potential to give slightly different information,” noted Dyson.
Advantages
MRI might be able to give far more detailed information regarding an injury than other imaging modalities such as X-ray and ultrasound.
Consider the proximal suspensory as an example. MRI can indicate how much of the injury is related to the bone versus the ligament and rule out damage to adjacent structures.
“Proximal suspensory (ligament versus enthesis) is a great reason for using MRI,” said Wulster Bills. “It can also be combined with PET because sometimes the MRI underestimates the degree of bone pain/pathology. PET can be great for showing earlier remodeling than what is seen on MRI alone.”
Disadvantages
“Knowledge of how to interpret images is certainly a limiting factor for MRI right now,” advised Dyson.
To interpret the images and determine what is or isn’t normal requires a highly detailed knowledge of anatomy. Movement artefacts, magnetic susceptibility artefacts (e.g., caused by a residual hoof nail clench), limited ability to interpret structures on the periphery of the area examined because of poorer image quality, etc., all limit image interpretation.
“Both performing and reading the images requires a lot of experience and knowledge,” said Dyson. “The skill of the person acquiring the images, the temperament of the horse and its response to sedation all influence image quality and therefore what can be learned from the images.”
MRI might be more expensive than radiographs, ultrasound or CT, especially if follow-up imaging is required.
“However, the costs of each diagnostic will depend on pricing structure, how capital costs of equipment are recovered, the throughput of scans, how efficient the operators are, etc.,” said Dyson. “Costs are a potential drawback for any imaging modality.”
Patient size limits this imaging modality to only the distal limb, and movement can greatly affect image quality. For image quality purposes, even the slight movement from a sedated horse swaying can have a great effect.
“Also, the higher up the limb that you go the more difficult it is in a standing horse to acquire images of diagnostic quality because of movement,” explained Dyson. “Think of a swinging pendulum. The carpus is like the bottom of the pendulum, with the foot being reasonably fixed in position.”
“Another disadvantage of MRI is that cartilage lesions cannot be reliably identified with low-field MRI,” said Wulster Bills.
Nuclear Scintigraphy
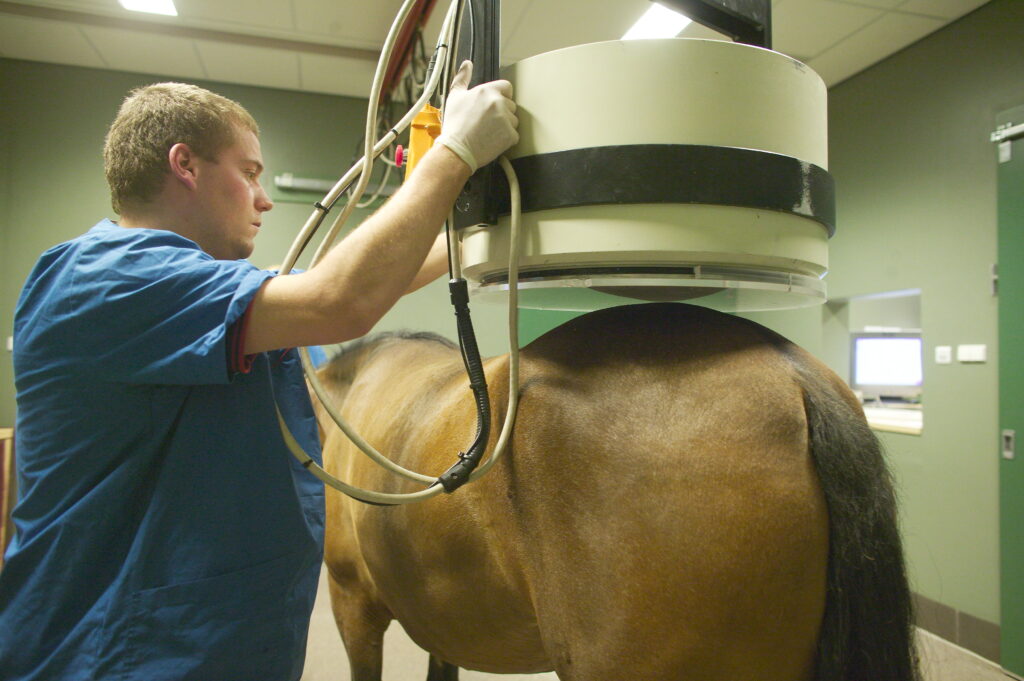
This advanced imaging technique involves intravenously injecting patients with a radioactive isotope such as 99-m technetium-labeled methylene diphosphonate (Tc99m-MDP). This molecule localizes to bone in proportion to osteoblastic activity (i.e., sites of bony remodeling). A gamma camera detects the radioisotope and creates images of the horse’s body with “hot spots” where the isotope is absorbed.
Common conditions diagnosed in horses via nuclear scintigraphy include bony lesions such as stress fractures in racehorses or enostosis-like lesions in sport horses.
“But really, all disciplines can benefit from scintigraphy if used appropriately,” said Wulster Bills.
Advantages
“This is the only modality that allows imaging of the whole body,” said Mathieu Spriet, DVM, MS, DACVR, DCVDI, Professor of Diagnostic Imaging at the University of California-Davis’ School of Veterinary Medicine. “It remains the most sensitive technique to diagnose stress fractures of the humerus, tibia and pelvis, as other imaging modalities typically cannot be used for imaging the proximal part of the limbs.”
This can potentially be used as a screening tool when patient behavior limits diagnostic anesthesia and for cases where inconsistent or multi-limb lameness are present.
“Scintigraphy can also be used to help determine the current clinical significance or activity of a lesion identified on radiographs,” added Wulster Bills.
Disadvantages
Scintigraphy as a screening might not always yield clear results.
A study by Quiney et al. (2018) examined the accuracy of skeletal scintigraphy as an “indiscriminate screening test in sports horses.” Horses undergoing scintigraphy that had a definitive diagnosis were included, and scintigraphy findings (i.e., radiopharmaceutical uptake) were judged as either relevant or non-relevant. Kappa statistics were then used to assess agreement between test results and final diagnosis.
“In total, there were 1,222 positive clinical diagnoses in 480 horses,” said Dyson. “There were 687 regions (56.4%) with no associated increased radiopharmaceutical uptake and 99 regions (8.1%) had non-relevant increased radiopharmaceutical uptake.”
Further statistical analyses revealed that “Scintigraphic examination is unlikely to lead to a full and correct diagnosis of the cause(s) of lameness or poor performance in sports horses when used as an isolated or indiscriminate screening tool.”
Wulster Bills added, “This modality is not inexpensive, requires the horse receive a significant amount of sedation because the procedure takes a few hours, and the horse must remain overnight at the hospital because it is radioactive.”
PET in Equine Patients
This is a cross-sectional imaging modality similar to nuclear scintigraphy in that the patient is injected with a radiotracer. But unlike scintigraphy that obtains a two-dimensional image, PET acquires three-dimensional data, allowing for three-dimensional rendering or visualization of two-dimensional cross-sectional images in any plane.
“That image is considered a ‘functional image’ because PET provides information on changes occurring at the molecular level that might precede structural changes,” explained Spriet.
“We are looking at the distribution of a radiotracer that accumulates at sites of molecular activity, sites of bone turnover, or glucose consumption,” he added. “The concept is that we are not looking at ‘size and shape’ (i.e., structural information) like with classic imaging techniques, but rather ‘physiological processes’ happening at the molecular level (i.e., functional information).”
For soft-tissue information, the radiotracer 18F-fluorodeoxyglucose (FDG) is used, whereas 18F-sodium fluoride (18F-NaF) is used for bone remodeling
Spriet and colleagues first used PET in 2015 to image horses with foot pain. From that study, interest in this imaging modality rapidly grew. Now, PET scanning is conducted by Spriet’s group at UC Davis as well as at the Santa Anita Park Thoroughbred racetrack. There are an additional eight scanners available throughout the U.S., including New Bolton Center (University of Pennsylvania), Rood and Riddle Lexington, Rood and Riddle Wellington, University of Florida World Equestrian Center, Ocala Equine Hospital, Kentucky Equine Hospital, Churchill Downs (operated by Hagyard), and Alamo Pintado Equine Hospital.
“The first installation of a PET scanner outside of the USA will be in the fall of 2023 at Melbourne University with Racing Victoria,” Spriet said.
In Spriet’s hands, PET imaging is primarily used for the following three reasons:
- As an alternative to scintigraphy for a cross-sectional bone scan of the distal limb;
- In addition to CT or MRI to distinguish active from inactive lesions and to detect early lesions not seen on CT or MRI. “Subchondral bone injuries in the early stage will be visible on PET as soon as the molecular process of bone turnover has started. MRI and CT will only show these lesions when sufficient demineralization has occurred,” said Spriet. Examples of such lesions are provided in Spriet’s 2022 AAEP presentation (available in the Convention Proceedings).
- To complement routine imaging obtained via radiographs or ultrasonography. That is, used as first line advanced imaging in order to decide whether CT or MRI is indicated and in which area.
Ideal ways to integrate PET into the diagnostic imaging process are either after CT and MRI (as mentioned above) or as a triage modality.
“Because PET can be performed quickly in the standing horse from foot to carpus or from foot to tarsus, it can help decide which area should be imaged with MRI—which is much more time consuming and expensive,” advised Spriet.
To date, the equine fetlock is the most commonly imaged region using PET, and those are done primarily in racehorses. But the foot, carpus, tarsus and origin of the suspensory ligament are also often imaged using this technique.
Spriet said that while most studies have focused on racehorses, he and his team have studies on sport horses scheduled for upcoming publication.
“In sport horses, we commonly image foot/fetlock combinations in forelimbs and fetlocks and tarsi in the hind limbs,” he said. “Imaging the suspensory is helpful in both fore- and hind limbs in these horses.”
Advantages
PET studies identify more lesions than scintigraphy, and a grading system quantifying the severity of a lesion can be used based on standardized uptake values (SUV). This grading system can help dictate how much time off a horse requires. In addition, follow-up PET scanning to sequentially measure SUV can be performed to help guide decisions regarding return to training, and to monitor possible recurrence when back in training.
Disadvantages
This imaging modality is still relatively limited in availability. Further, only the distal limb can be imaged. Any functional imaging of the proximal limb or axial skeleton must be conducted using nuclear scintigraphy.
Integrating Advanced Imaging into Lameness Examinations

When using advanced imaging in lameness cases, a full physical examination and nerve blocking are usually essential first steps to localize the source(s) of pain.
As mentioned above, it is not usually practical to scan multiple areas or entire horses using any of the advanced imaging modalities. Scans can take time, especially if both the lame and non-lame limbs are being scanned and compared. Thus, having the patient anesthetized or even sedated sufficiently for prolonged periods is not often reasonable. Further, imaging lengthy regions of the horse’s limbs will add to the overall expense of the procedure.
“Additionally, we can find lesions in many locations that may not be relevant to the horse’s current cause of lameness, and this is where blocking is absolutely necessary to determine clinical significance,” said Wulster Bills.
Thus, the primary practitioner should attempt to localize the source(s) of pain to the best of his/her ability before recruiting the power of radioisotopes, magnetic fields or radiation. Admittedly, this can be challenging considering that perineural and intra-articular anesthetics can diffuse beyond the textbook boundaries, obscuring precise localization.
Which Advanced Imaging Modality to Use for Horses?
So, which modality should you choose for lameness localized to a particular region, but a firm diagnosis has not been achieved?
Some of us might have the impression that the following flow chart is how advanced imaging fits into clinical practice, where CT is the gold standard for bone, MRI for soft tissues in the foot, nuclear scintigraphy for shins, and PET for a racehorse’s ankles.
However, lameness cases rarely follow the textbook or fall into a nice tidy paradigm that will follow this flow.
For example, Dyson noted that “Bone lesions characterized by increased signal intensity in STIR images may not be apparent on CT images.”
Wulster Bills said that if radiographs and ultrasound have not identified a cause of lameness, she considers cross sectional imaging and uses the following categories:
- Where the primary concern is bone (e.g., subchondral bone injury);
- Where the primary concern is soft tissue; or
- Cases in which there are concerns about both bone and soft tissue.
“I also question whether the lameness is in a single region or if the horse has partial improvement with multiple blocks,” noted Wulster Bills. “If the latter is true, then we may actually do PET next, using a dual tracer for both soft tissue and bone rather than just one isotope, depending on greatest clinical concern.”
As one example of the value of MRI in the equine foot, Dyson said, “Increased signal intensity in STIR images in the spongiosa of the navicular bone represents one form of navicular disease that cannot be detected by other imaging techniques. If this reflects bone edema and necrosis (mediated by increased osteoclastic activity), this will not be detected by radiography, scintigraphy or CT.”
Ultimately, however, the decision regarding the imaging modality of choice often needs to be made depending on what modality is available in the area.
Based on the current body of evidence regarding advanced imaging in lameness evaluations, Dyson makes the following observations:
- PET is still relatively in its infancy in sport horses. “It is an extremely sensitive technique for picking up abnormal bone activity in racehorses,” Dyson said.
- Scintigraphy is more likely to be used in sports horses if a proximal limb injury is suspected.
- In racehorses, where stress-related bone injury is so common and often present in more than one location, either scintigraphy or PET would be applicable. “Again, you would only use CT or MRI if you had localized pain causing lameness by diagnostic anesthesia so you knew where to scan. In a poorly performing racehorse lame in all four limbs you would use PET or scintigraphy,” explains Dyson.
- Scintigraphy allows you to examine a much larger area of a horse, both forelimbs for example, whereas MRI is used to focus on a specific area (from the carpus/ tarsus and below) to which pain has been first localized by diagnostic anesthesia. “So, for example, if lameness was abolished by palmar digital nerve blocks and radiographic examination yielded negative results, then MRI would probably be the imaging modality of choice,” noted Dyson. “But we do need to bear in mind that a palmar digital block could influence pastern pain or occasionally fetlock region pain. Thus, it would take three sets of MRI scans—the foot, pastern and fetlock—to image those regions properly if using the low-field Hallmarq system. The high-field systems have larger fields of view, so only two sets are needed, one for the foot and pastern combined and another of the fetlock. This obviously would be time consuming and expensive.”
- MRI is the current gold standard for soft-tissue injuries; however, similar information can be acquired from contrast CT studies using soft tissue algorithms where available.
- As standing CT has developed, it is being used increasingly because images can be acquired so quickly with good image resolution.
Additionally, Dyson said that our knowledge about the perceived limitations of CT and MRI is increasing, breaking down existing stereotypes.
“Dr. Annamaria Nagy and I are conducting some studies at the University of Budapest, Hungary, comparing low-field MRI and CT,” she said. “It is remarkable in some horses what is missed with MRI and seen on CT and vice versa.”
Even with all these diagnostic options, a clear answer for the lameness might continue to elude us.
“In some cases, when a definitive diagnosis cannot be achieved, it might be necessary to go back and re-block the horse to more precisely localize the lesion,” said Wulster Bills. “In fact, we often use the lesions we have found on advanced imaging to do more targeted blocking (e.g. intra-articular pastern).”
Further, don’t forget that advanced imaging isn’t always required!
Dyson said, “If a horse had an acute onset lameness that blocked out 100% to a subcarpal block and ultrasonographic images showed structural changes of the proximal aspect of the suspensory ligament that were consistent with the degree of lameness, I would not go further. If the degree of lameness was not explained by the ultrasonographic images, then I would consider the use of MRI, CT or scintigraphy to determine the presence of bone injury at the enthesis or to identify another injury contributing to pain and lameness.”
Final Thoughts on Advanced Imaging Options for Horses
The goal of advanced imaging is to obtain a diagnosis that is as accurate as possible so that the appropriate treatment can be initiated. Ideally, a clear prognosis would also be given.
“These modalities are so sensitive it is now possible to pinpoint the exact anatomic location of the injury,” noted Wulster Bills. “Where traditional X-rays may suggest navicular disease, MRI and CT can better define the problems, such as navicular bone degenerative changes, a core lesion of the deep digital flexor tendon, distal interphalangeal joint osteoarthritis, collateral ligament desmopathy and/or distal sesamoidean ligament desmopathy.”
Indeed, the imaging modalities we now have and continue to develop are powerful. Possibly too powerful in some situations.
“The more we use advanced imaging the more abnormalities we find,” said Dyson. “And determining the current or future clinical significance is not necessarily straightforward. A radiologist may provide a list of abnormalities identified, but these have to be placed into the clinical context by the practitioner, which is not always an easy feat.”
When consulting with a radiologist, Wulster Bills said, “A detailed clinical history and blocking pattern provided to the radiologist are essential for the best interpretation. Taking a list of abnormalities from the radiologist and having the practitioner try to apply it does not always work. For instance, I may say a horse has moderate chronic collateral ligament desmopathy, but based on other findings, results of blocking and degree of lameness, I may not think this is the primary cause of lameness despite it being the highest ‘grade’ of injury. Radiologists will also mention when we think we have not found the cause of lameness because the findings are insufficient to cause the reported degree of lameness, in our opinion.”

![[Aggregator] Downloaded image for imported item #18392](https://s3.amazonaws.com/wp-s3-equimanagement.com/wp-content/uploads/2025/09/30141455/EDCC-Unbranded-7-scaled-1-768x512.jpeg)
![[Aggregator] Downloaded image for imported item #18379](https://s3.amazonaws.com/wp-s3-equimanagement.com/wp-content/uploads/2025/09/30140703/EDCC-Unbranded-15-scaled-1-768x511.jpeg)

