This article originally appeared in the Fall 2025 issue of EquiManagement. Sign up here for a FREE subscription to EquiManagement’s quarterly digital or print magazine and any special issues.

According to multiple sources, interleukin-1 receptor agonist protein (IRAP) therapy, also referred to as autologous conditioned serum (ACS), plays a prominent role in treating musculoskeletal conditions in horses. Wilson et al. (2018) reported that 81.4% of veterinarians responding to a survey about which therapy they used for injury or performance issues relayed using IRAP. Similarly, in 2021 Zanotto and Frisbie surveyed American Association of Equine Practitioners members and reported that 83.3% of survey respondents said they used ACS as a joint therapy. Della Tommasa et al. (2023) reported a “notable” increase in ACS utilization in recent years.
Despite its popularity, only six studies (meeting inclusion criteria for their systematic review) on ACS exist, said Della Tommasa et al., only one of which was a controlled study. With this in mind, let’s delve into the world of ACS.
Producing and Quantifying ACS
In 2003, researchers (Frizziero et al. and Meijer et al.) reported that incubating samples of whole blood with specialized beads (e.g., coated in chromium sulfate or borosilicate) increases white blood cell (primarily monocyte) production of IRAP and other anti-inflammatory cytokines. That serum, they said, could then be used to treat various inflammatory and degenerative conditions, most notably equine osteoarthritis (OA).
“The rationale is that IRAP competes with the pro-inflammatory cytokines interleukin (IL)-1 alpha and beta. In OA, IL-1 beta is of particular interest because it is produced by synoviocytes and chondrocytes and is a central driver of articular inflammation,” relays Kyla Ortved, DVM, PhD, DACVS, DACVSMR, an associate professor of large animal surgery from the University of Pennsylvania’s New Bolton Center.
Ortved explains that inflammation within the joint starts a cascade of events that increases the production of matrix metalloproteinases (MMPs) and other degradative enzymes (e.g., aggrecanases). These enzymes degrade the cartilage’s extracellular matrix, specifically the proteoglycans and type II collagen, leading to a progressive loss of cartilage function.
In addition to IL-1Ra, ACS contains other anti-inflammatory molecules and immunomodulators, such as soluble tumor necrosis factor receptor, insulin-like growth factor-1 (IGF-1), and interleukin (IL)-10.
“But IRAP also contains some pro-inflammatory molecules, including IL-1 beta and tumor necrosis factor-alpha, as do other autologous blood-based products” says Ortved.
The rationale is the anti-inflammatory mediators outweigh the effects of the pro-inflammatory mediators.
Treating Horses With ACS

After incubating whole blood with the specialized beads for 24 hours at 37 degrees C (98.6 F), the conditioned serum is separated and collected sterilely into syringes.
The most common indication for IRAP is acute synovitis and mild to moderate osteoarthritis. A typical treatment protocol, says Ortved, involves anywhere from two to five injections administered seven to 10 days apart. She says this approach stems from the original protocol used in initial proof of concept research studies.
“The volume to be injected is based off joint volume,” she explains. “For example, we would use 1 to 2 milliliters in small joints like the distal tarsal joint but 8 milliliters for the femoropatellar joint.”
Any remaining IRAP can be frozen at -20 C (-4 F), which is standard fridge freezer temperature.
Adverse Events Following IRAP
Because IRAP is not FDA-approved (it is classified as a medical device), safety data is not centrally reported like it is for medications. Little data regarding safety and adverse effects are available in the literature.
“I don’t think the incidence of joint flares is any different than other intra-articular therapy,” says Ortved. “So, they can theoretically occur but would only rarely.”
Variability in ACS
Because ACS is produced from each individual horse, it’s not surprising that the conditioned serum varies from one patient to another.
“ACS may be regarded as a ‘soup’ containing a myriad of potential bioactive factors, and we have yet to elucidate either the full content of ACS or the full array of potential treatment effects,” relays Cathrine Fjordbakk, DVM, PhD, DECVS, DECVSMR, Professor in Equine Surgery, Norwegian University of Life Sciences, in Ås. “Characterizing ACS content is both costly and time-consuming, not to mention that we’re limited to testing known factors for which there are available tests.”
To complicate the matter, Fjordbakk says ACS also contains significantly more exosomes than nonincubated serum (Buchheit et al., 2023), of which we have very little knowledge.
“In fact, in human medical literature, ACS was recently renamed as the ‘blood cell secretome’ (Shirokova et al., 2020), reflecting the breadth of potential bioactive agents released by the coagulation cascade and thermal induction that takes place during incubation,” says Fjordbakk. “On top of this, all researchers investigating ACS content tell us there are massive interindividual variations in all cytokines and growth factors we currently are able to test for. This not only complicates ACS research but also demonstrates one major problem with orthobiologics: We really don’t know the specific content and quality of the product we’re using for any particular patient.”
One factor that does appear to affect ACS quality is inflammation.
Fjordbakk and her associates published a study in 2015 that involved preparing ACS pre- and postoperatively in a cohort of 15 stallions castrated under general anesthesia. Based on postoperative serum amyloid A (SAA) levels, horses were categorized as having responded to the surgery and anesthesia with mild, moderate, or marked surgical stress.
“We found that IGF-1 was significantly reduced in all postoperative ACS samples, and IL-1Ra and TGF-beta were significantly reduced in postoperative ACS samples of horses in the marked surgical stress group only,” she explains. “We interpreted this as a negative effect of the ACS profile, under the assumption that these cytokines and growth factors indeed may have beneficial effects in the joint.”
She therefore recommends avoiding drawing blood for ACS preparation postoperatively—for example, from horses undergoing arthroscopy since this procedure also triggers elevated SAA for up to five days postoperatively.
“I also make sure the horse is systemically healthy without any concurrent inflammatory conditions going on,” she adds.
Finally, a synergistic effect between different components of the ACS might be at play.
“We’ve demonstrated a significant association between the ACS content of IGF-1 and IL-1Ra and clinical response (Marques-Smith et al., 2020),” Fjordbakk says. “In that study, we included 20 low-grade lame harness racehorses, all of which were treated with three ACS injections approximately two weeks apart. Two weeks after the last treatment, 58% of the horses were no longer lame, whereas lameness was unchanged in the rest of the horses. Both IGF-1 and IL-1Ra content were significantly higher in ACS from the responder group, leading us to conclude that therapeutic benefit of ACS treatment may be related to these two factors.”
Contraindications for ACS
Aside from the potential for joint flare or joint infection inherent to any intra-articular injection, few known adverse events from ACS exist.
Ortved and others have, however, wondered if a horse is receiving medications, will that drug be present in the conditioned ACS product? To explore this potential effect further, Ortved et al. (2017) administered 0.1 mg/kg firocoxib orally to six healthy mares before their morning meal. She administered two doses 24 hours apart, collecting blood samples four hours following the second dose to measure firocoxib levels in both the blood and the ACS product (produced using a commercial kit). ACS samples remained frozen until time of use, which was after the horses tested negative for firocoxib in their bloodstream. After administering the firocoxib-containing ACS intra-articularly, Ortved collected blood samples intermittently for 48 hours to measure serum firocoxib levels.
Firocoxib levels in the serum and ACS four hours after the second dose were 33.3 +/- 4.27 ng/mL and 35.4 +/- 4.47 ng/mL. After that, firocoxib levels in the horses’ serum remained below detection level at all time points.
Whether longer-term NSAID administration would change these results or whether other types of medications can be present in ACS remains unknown.
Kit-less ACS?
Despite all the time, effort, and money put into studying the specialized glass beads to activate monocytes in whole blood, research by Fjordbakk et al. (2023) and other groups suggests commercial kits and specialized beads might be unnecessary. Instead, veterinarians might be able to produce ACS using other vessels, such as standard sterile 50-milliliter plastic centrifuge tubes.
“In our 2015 surgical stress study mentioned above, we noticed a significantly higher content of IL-1Ra and IGF-1 when blood was incubated in plain red-top vacutainer tubes versus two different specialized ACS containers with beads,” Fjordbakk explains.
This finding corroborated results reported by Hraha et al. in 2011.
“Since those studies, some veterinarians have used the vacutainer tube method as an economic way of producing ACS,” Fjordbakk adds. “However, drawing conditioned serum from multiple 10-milliliter vacutainer tubes, which is necessary to procure enough volume for a regular ACS treatment course, would drastically increase the handling steps compared to handling one single 55-milliliter ACS container.”
Therefore, although the desired cytokine profile was superior in serum incubated in the vacutainer tubes, Fjordbakk and colleagues did not recommend using the vacutainer technique because they felt it posed a safety risk.
“This finding did, however, spark our most recent study (Nakken et al., 2023), where we wanted to test a large sterile container without beads against a commercially available ACS kit,” relays Fjordbakk.
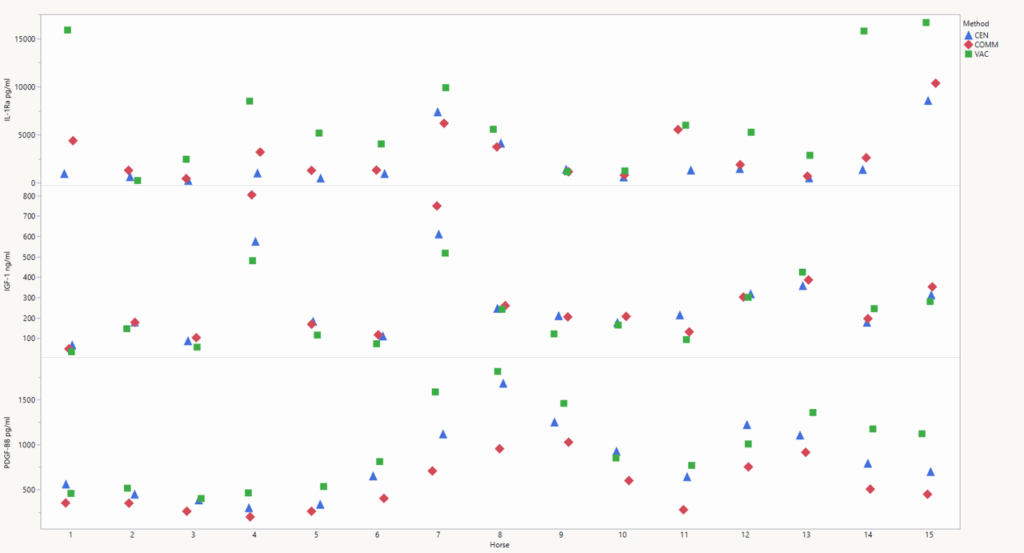
In that 2023 study, the goal was to determine the level of various cytokines and growth factors in ACS produced using three different methods: a commercial kit, 50-milliliter centrifuge tubes, and 10-milliliter serum vacutainers. The team collected 131 milliliters of blood from 15 healthy horses and transferred it into each of the three types of tubes. All tubes were incubated at 37 C for 22 to 24 hours and the conditioned serum separated. Those samples were analyzed for IL-1Ra, IL-10, IL-1 beta, TNF-alpha, TGF-beta, platelet-derived growth factor-BB, and IGF-1.
They noted no significant differences in either IRAP or IGF-1 between the ACS produced by the commercial kit and the plain 50-milliliter centrifuge tubes. Platelet-derived growth factor-BB (PDGF-BB) was significantly higher in the ACS produced by the 50-milliliter centrifuge tubes than the commercial kit.
The team also noted that the vacutainer was the superior method of increasing ACS content of IL-1Ra and PDGF-BB. These levels were significantly higher in vacutainers versus the larger containers.
However, the multiple handling steps involved in vacutainer use can potentially “increase the risk of breaching aseptic technique,” they reiterated.
Evidence of Efficacy
As noted, Della Tommasa et al. are unimpressed with the body of literature examining the efficacy of ACS in horses with OA. They included only six studies in their systematic review of clinical data. All included studies used different protocols for ACS injection, and only one study included histologic examination of joint tissue (Frisbie et al., 2007).
That study is the only randomized controlled trial published to date and likely provides the most powerful data we have. It involved a carpal chip model, which is the closest representation of traumatic joint inflammation available to study naturally occurring joint disease. The researchers injected either ACS or a placebo into a middle carpal joint after inducing OA via chip fracture. They administered treatments on Days 4, 21, 28, and 35. They observed significant improvement in lameness in horses treated with ACS and decreased synovial membrane hyperplasia in ACS-treated horses compared with placebo-treated joints. Further, the synovial fluid concentration of IRAP increased following treatment with ACS.
“We definitely need more information on responders and nonresponders as well as inter- and intrahorse variability,” says Ortved.
Fjordbakk adds, “A stallside test for the ACS content of IL-1Ra and IGF-1 could potentially enable us to identify responders from nonresponders prior to starting treatment.”
Ortved and Fjordbakk agree that current treatment protocols are merely empirical, both when it comes to treatment interval and treatment volume.
The Final Word
In sum, limited controlled studies show this therapy’s efficacy, we don’t know what’s in ACS or how it truly works, and we don’t know why some horses respond while others do not.
“I think we need to realize that the optimal treatment of equine OA is still not identified, and there are pros and cons of every single treatment out there,” says Fjordbakk. “For instance, we are getting more and more evidence that repeated steroid injections may have detrimental effects, in essence only masking clinical signs although the joint pathology is still progressing. My reasoning for choosing orthobiologics is the apparent safety of the products, both in the short and long term.”
Related Reading
- From a Whisper to a Bang: Where We Are With Orthobiologics
- Healing With Horsepower: Recent Orthobiologics Research
- Daily Vet Life: Successful IRAP Outcomes
Stay in the know! Sign up for EquiManagement’s FREE weekly newsletters to get the latest equine research, disease alerts, and vet practice updates delivered straight to your inbox.




